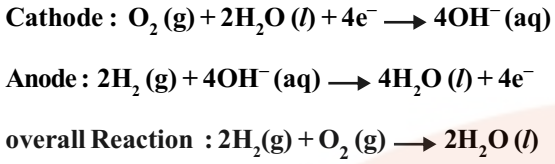Fuel Cells
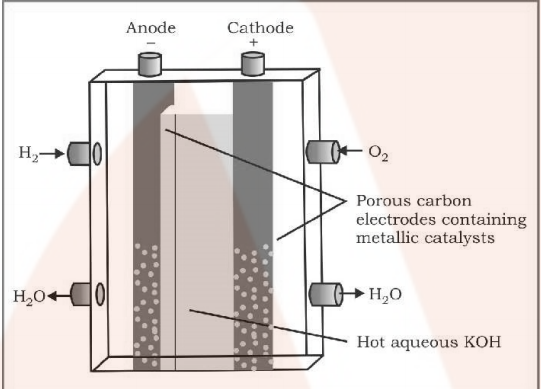
A fuel cell differs from an ordinary battery in the sense that the reactants are not contained inside the cell but are externally supplied from an external reservoir. A fuel cell is used in space vehicles and in this cell the two gases are supplied from external storages. In this cell, carbon rods are used as electrodes with KOH as the electrolyte.