Heat Engine and efficiency
A heat engine is a device by which a system is made to undergo a cyclic process that results in the conversion of heat to work.
For any heat engine, there are three essential requirements.
- SOURCE: A hot body at fixed temperature T1 from which heat engine can draw heat
- SINK: A cold body, at a fixed lower temperature T2, to which any amount of heat can be rejected.
- WORKING SUBSTANCE: The material, which on being supplied with heat will do mechanical work.
Working Substance:
- In the heat engine, working substances could be gas in a cylinder with a moving piston.
- In the heat engine, working substance takes heat from the source, converts a part of it into mechanical work, gives out rest to the sink and returns to the initial state. This series of operations constitutes a cycle.
- This cycle is represented in fig below
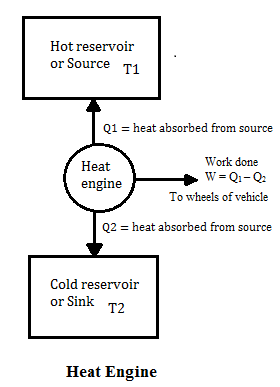
- Work from heat engine can be continuously obtained by performing same cycle again and again.
- Consider,
Q1 = heat absorbed by working substance from source
Q2 = heat rejected to the sense
W = net amount of work done by working substance
Q1 – Q2 = net amount of heat absorbed by working substance
∆U = 0 since in the cycle Working substance returns to its initial condition.
So on application of first law of thermodynamics
Q1 – Q2 = W
-
Thermal efficiency of heat engine

From this equation it is clear that
Q = 1 for Q2 = 0
And there would be 100% conversion of heat absorbed into work but such ideal engines are not possible in practice.
Refrigerators and Heat Pumps
- Refrigerators work in reverse direction of heat engines.
- In refrigerators working substance extracts heat Q2 from sink at lower temperature T2
- Some external work is performed by the compressor of the refrigerator and then heat Q1 is rejected to the source, to the radiator of the refrigerator.
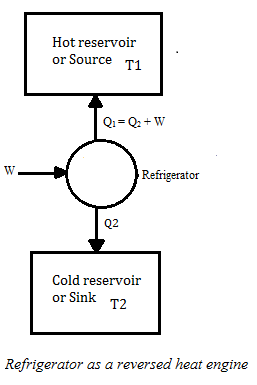
The coefficient of performance:
β= Amount of heat absorbed from the cold reservoir/work done in running the machinery
Q2 – heat absorbed from cold reservoir.
Q1 – heat rejected to hot reservoir during one complete cycle
W = (Q1-Q2) is the work done in running the machinery
thus,
![]()
- Like heat engines, refrigerators cannot work without some external work done on the system. Hence coefficient of performance cannot be infinite.

