
Physical Properties of ethers:
Miscibility:
Miscibility of ethers with water resembles those of alcohols of the same molecular mass. This is due to the fact that just like alcohols, oxygen of ether can also form hydrogen bonds with water molecule.

Boiling points:
Ethers have much lower boiling points than alcohols. This is due to the presence of hydrogen bonding in alcohols. Hydrogen bonding is absent in ethers.
Chemical Properties of ethers:
Cleavage of C–O bond in ethers:
R-O-R’ + HX → R-X + R’OH
The order of reactivity of hydrogen halides is as follows: HI >HBr>HCl
Alkyl halide formed is always the lower alkyl group. But if a tertiary alkyl group is present, the alkyl halide is always tertiary. In case of phenolic ethers, the cleavage occurs with the formation of phenol and alkyl halide.
Electrophilic substitution:
The alkoxy group (-OR) is ortho, para directing and activates the aromatic ring towards electrophilic substitution in the same way as in phenol.

Halogenation:
Phenylalkyl ethers undergo usual halogenation in the benzene ring, e.g., anisole undergoes bromination with bromine in ethanoic acid even in the absence of iron (III) bromide catalyst. It is due to the activation of benzene ring by the methoxy group. Para isomer is obtained in 90% yield.

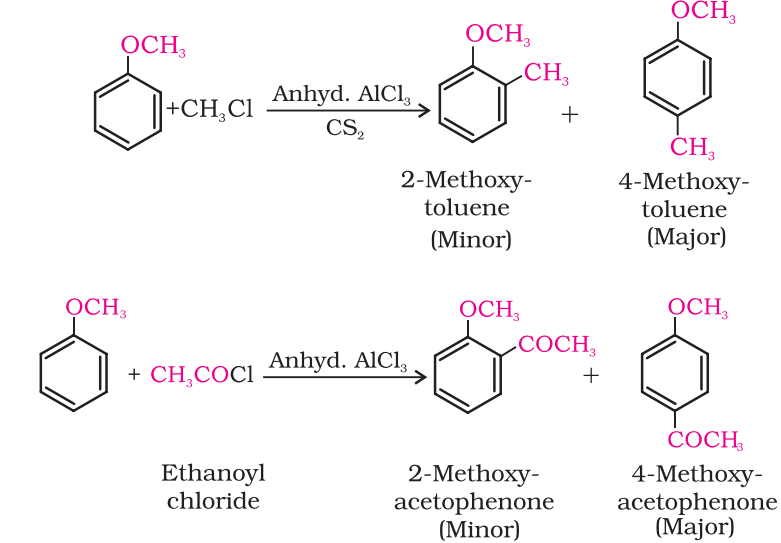
Friedel-Crafts reaction:
Anisole undergoes Friedel-Crafts reaction, i.e., the alkyl and acyl groups are introduced at ortho and para positions by reaction with alkyl halide and acyl halide in the presence of anhydrous aluminum chloride (a Lewis acid) as catalyst.
Nitration:
Anisole reacts with a mixture of concentrated sulphuric and nitric acids to yield a mixture of ortho and para nitroanisole.


