
Proteins
The word protein is derived from Greek word, “proteios” which means primary or of prime importance. All proteins are polymers of -amino acids.
Amino Acids
The compounds containing amino group (-NH2) and carboxylic group (-COOH) are called amino acids. The general structure is:
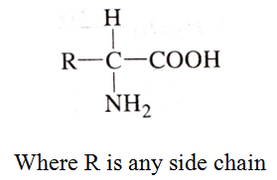
Classification of Amino Acids
- Amino acids are classified as acidic, basic or neutral depending upon the relative number of amino and carboxyl groups in their molecule.
- The amino acids, which can be synthesised in the body, are known as non-essential amino acids.
- On the other hand, those which cannot be synthesised in the body and must be obtained through diet, are known as essential amino acids.
Structure of protein
Peptide Bond
Peptide linkage is an amide linkage formed by condensation of two amino acids involving −NH2 group of one amino acid and the −COOH group of the other amino acid with the loss of water.
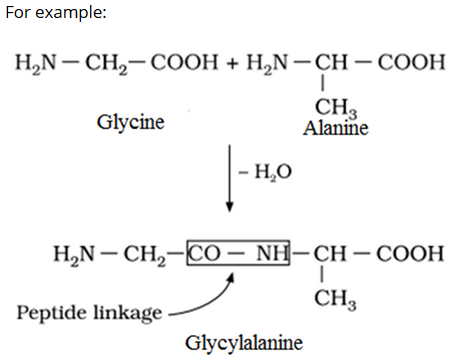
On the basis of number of amino acids undergoing the condensation, the peptides can be classified as:
- Dipeptide: It is a peptide composed of two amino-acid residues.
- Tripeptide: It is a peptide composed of three amino-acid residues.
- Polypeptide: It is a peptide composed of more than ten amino-acid residues.
Protein
Peptide having molecular mass higher than 10,000 u is called a protein.
Classification of Proteins
On the basis their molecular shape, proteins can be classified as:
- Fibrous proteins: The polypeptide chains run parallel and are held together by hydrogen and disulphide bond forming a fibre like structure. They are generally insoluble in water. For example: Keratin (present in hair, wool, silk) and myosin (present in muscles), etc.
- Globular proteins: The polypeptide chains coil around to give a spherical shape. These are soluble in water.
- For example: Insulin and albumins.
Structure and shape of proteins can be studied at four different levels, i.e., primary, secondary, tertiary and quaternary, each level being more complex than the previous one.
Primary Structure of proteins:
The sequence of amino acids is said to be the primary structure of a protein.

Secondary Structure of proteins:
This structure of a protein refers to the shape in which a long polypeptide chain can exist.

There are two secondary structures of proteins:
- α-Helix: In α-Helix, the polypeptide chain coil itself into a right-handed screw with the −NH group attached to the −C=O of an adjacent turn of the helix by the hydrogen bonding. It generally exists when R- group is large.
- β-Helix: In this structure, the peptide chains are stretched out to nearly maximum extension and then laid side by side, held together by intermolecular hydrogen bonds. It generally exists when R- group is small.
Tertiary Structure of proteins:
The tertiary structure of proteins represents overall folding and superimposition of the polypeptide chains, i.e., further folding of the secondary structure giving rise to a ball shape or spheroidal shape structure.
It is stabilized by covalent, ionic, hydrogen and disulphide bonds.

Quaternary structure:
Some of the proteins are composed of two or more polypeptide chains referred to as sub-units. The spatial arrangement of these subunits with respect to each other is known as quaternary structure.
Denaturation of Proteins
- The most stable conformation of a protein at a given temperature and the pH is known as its native state.
- The loss of biological activity of proteins when a protein in its native form, is subjected to physical change like change in temperature or chemical change like change in pH, is called denaturation of protein.
- For example: Coagulation of egg white on boiling, curdling of milk which is caused due to the formation of lactic acid by the lactobacillus bacteria present in milk.

