
Stoichiometric Defects
These are the point defects that do not disturb the stoichiometry of the solid. They are also called intrinsic or thermodynamic defects.
Basically these are of two types:
- Vacancy defects
- Interstitial defects.
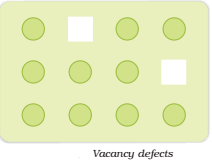
Vacancy Defects:
When some of the lattice sites are vacant, the crystal is said to have vacancy defect (Fig.). This results in decrease in density of the substance. This defect can also develop when a substance is heated.
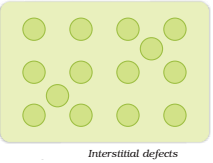
Interstitial Defect:
When some constituent particles (atoms or molecules) occupy an interstitial site, the crystal is said to have interstitial defect (Fig.). This defect increases the density of the substance.
Vacancy and interstitial defects as explained above can be shown by non-ionic solids. Ionic solids must always maintain electrical neutrality. Rather than simple vacancy or interstitial defects, they show these defects as Frenkel and Schottky defects.
Frenkel Defect:
This defect is shown by ionic solids. The smaller ion (usually cation) is dislocated from its normal site to an interstitial site (Fig.). It creates a vacancy defect at its original site and an interstitial defect at its new location. Frenkel defect is also called dislocation defect.

It does not change the density of the solid. Frenkel defect is shown by ionic substance in which there is a large difference in the size of ions, for example, ZnS, AgCl, AgBr and AgI due to small size of Zn2+ and Ag+ ions.
Schottky Defect:
It is basically a vacancy defect in ionic solids. In order to maintain electrical neutrality, the number of missing cations and anions are equal (Fig.).

- It occurs in ionic compounds
- It decrease the density of solid
- Shown by ionic compound having similar size of cation and anion.
For example, NaCl, KCl, CsCl and AgBr. It may be noted that AgBr shows both, Frenkel as well as Schottky defects.

