
Rutherford proposed a new model of the atom based on Alpha-Particle Scattering experiment.
In 1911, Ernst Rutherford suggested some experiments to H. Geiger and E. Marsden.
Both scientists performed an experiment whose schematic diagram is shown in the figure given.
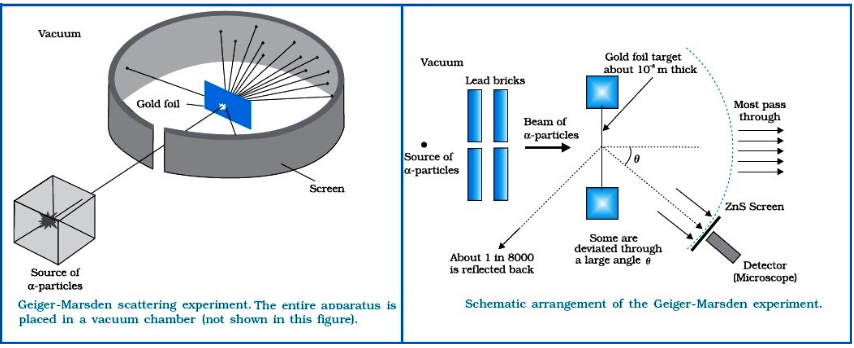
They directed a beam of 5.5 MeV α-particles emitted from a radioactive source at a thin metal foil made of gold.
The beam was allowed to fall on a thin foil of gold thickness 2.1 × 10-7 m.
The scattered alpha-particles were observed through a rotatable detector consisting of zinc sulphide screen and a microscope. The scattered alpha-particles on striking the screen produced brief light flashes or scintillations. These flashes may be viewed through a microscope and the distribution of the number of scattered particles may be studied as a function of angle of scattering
Observation of the Alpha (α) scattering Experiment
Many of the α-particle pass through the foil which means that they do not suffer any collisions. Approximately 0.14% of the incidents α-particles scatter by more than 10 and about 1 in 8000 deflect by more than 900.
Conclusion from Alpha (α) scattering Experiment
Based on α scattering experiment, Rutherford concluded the following important points.
According to him
- Scattering of alpha particle is due to columbic force between positive charge of α particle and positive charge of atom.
- Rutherford’s experiments suggested the size of the nucleus to be about 10–15 m to 10–14 m.
- The electrons are present at a distance of about 10,000 to 100,000 times the size of the nucleus itself.
- Atom has a lot of empty space and the entire mass of the atom is confined to very small central core also known as nucleus.
Some important terms related to Alpha (α) Scattering Experiment
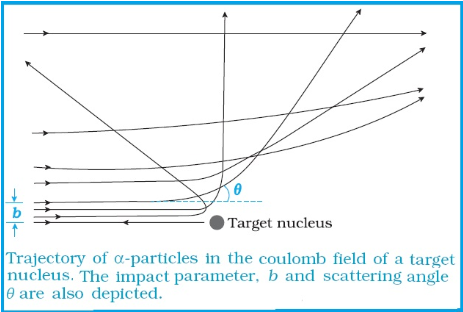
Impact Parameter:
It is the perpendicular distance of the velocity vector of α particle from the central line of the nucleus, when the particle is far away from the nucleus of the atom.
Angle of scattering:
It is the angle made by α particle when it gets deviated from its original path around the nucleus.

