Dielectrics
Dielectrics are non-conducting substances. In contrast to conductors, they have no or the negligible number of charge carriers. In a dielectric, free movement of charges is not possible. It turns out that the external field induces dipole moment by stretching or re-orienting molecules of the dielectric. The collective effect of all the molecular dipole moments is net charges on the surface of the dielectric which produce a field that opposes the external field.
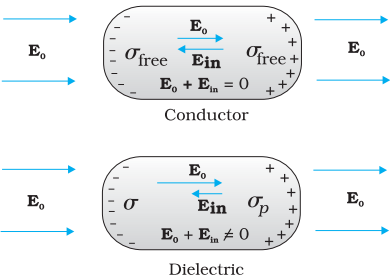
The difference in behavior of a conductor and a dielectric in an external electric field.
Types of dielectrics:
Dielectrics are two types:
-
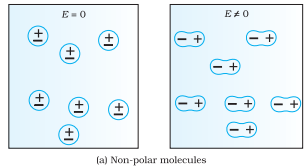
Non-Polar Dielectrics:
When the centre of positive charge coincides with the centre of negative charge in a molecule, e.g., Nitrogen, Oxygen, CO2 etc.
Polar Dielectrics:
When the centre of positive and negative charges do not coincide because of the asymmetric shape of the molecules, e.g., NH3, HCL etc.
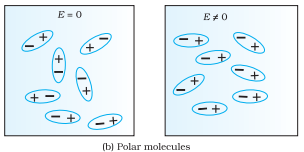
The non-polar molecule thus develops an induced dipole moment. The dielectric is said to be polarized by the external field. Substances for which this assumption is true are called linear isotropic dielectrics. The induced dipole moments of different molecules add up giving a net dipole moment of the dielectric in the presence of the external field.
Thus in either case, whether polar or non-polar, a dielectric develops a net dipole moment in the presence of an external field. The dipole moment per unit volume is called polarisation and is denoted by P. For linear isotropic dielectrics
![]()
where is a constant characteristic of the dielectric and is known as the electric susceptibility of the dielectric medium. It is possible to relate to the molecular properties of the substance.

