
Preparation of Dihydrogen, H2
Laboratory Preparation of Dihydrogen
- It is usually prepared by the reaction of granulated zinc with dilute hydrochloric acid.
![]()
- It can also be prepared by the reaction of zinc with aqueous alkali.
![]()
Commercial Production of Dihydrogen
- By the electrolysis of acidified water

- Form water gas (Bosch process)
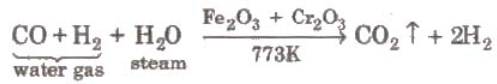
- From steam (Lane’s process) super-heated steam is passed over iron filings heated to about 1023-1073 k when hydrogen is formed.
- Highly pure (> 99.95 %) dihydrogen is obtained by electrolyzing warm aqueous barium hydroxide solution between nickel electrodes.
- From hydrocarbons by partial oxidation

- It is also obtained as a by-product in the manufacture of NaOH and chlorine by the electrolysis of brine solution.
During electrolysis, the reactions that take place are

Properties of Dihydrogen
Physical Properties
- Dihydrogen is a colourless, odourless and tasteless gas.
- It is a combustible gas.
- It is insoluble in water.
- It is lighter than air.
Chemical Properties of Dihydrogen
- Reaction with halogens: It reacts with halogens, X2 to give hydrogen halides. HX.

(F can react with hydrogen in dark also, iodine requires a catalyst)
- Reaction with dioxygen:

The reaction is highly exothermic.
- Reaction with dinitrogen: With dinitrogen to form ammonia

- Reaction with metals: Here H2 acts as oxidising agent.
![]()
Where (M = alkali metal)
- Reaction with metal ions and metal oxides: It reduces some metal ions in aqueous solution and oxides of metals (less active than iron) into corresponding metals

- Reaction with organic compounds: It reacts with many organic compounds in the presence of catalysts to give useful hydrogenated products of commercial importance.
- Hydrogenation of vegetable oils using nickel as catalyst gives edible fats (margarine and Vanaspati ghee).
- Hydroformylation of olefins yields aldehydes which further undergo reduction to give alcohols.

Use of Dihydrogen
- It is used in the manufacture of CH3OH.
- It produces temperature of 2850oC and oxy-atomic hydrogen flame produces a temperature of 4000oC, so it is used in oxy-hydrogen flame.
- The largest single use of H2 is in the synthesis of NH3 which is used in the manufacture of HNO3 and fertilizers.
- Liquid hydrogen (LH2) is used as rocket fuel.
- H2 is used as a reducing agent in extraction of metals.
- H2 is used in fuel cell for generating electrical energy.
- Hydrogen is used in the manufacture of synthetic petrol.
(By heating H2 with coal and heavy oils under very high pressure in the presence of catalyst).

