Magnetic Properties of Solid
Electrons are charged particles and it generates a magnetic field around itself. The magnetic field arises due to the spinning of electron at its own axis and movement of atomic orbital around nucleus.
Classification of magnetic properties:
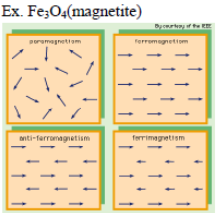
Paramagnetism:
- Weakly attracted by magnetic fields.
- Alignment of magnetic dipole in the same direction of magnetic field
- Loose magnetism in absence of magnetic field
- They have unpaired electrons
- Example: Cu2+, Fe3+
Diamagnetism:
- Weakly repelled by magnetic fields
- Alignment of magnetic dipole in the opposite direction of magnetic field
- They have all the electrons paired
- Example: H2O, NaCl
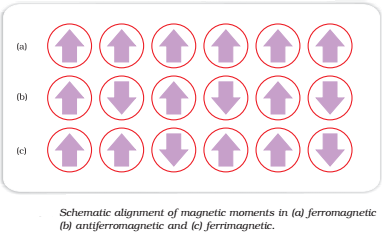
Ferromagnetism:
- Strongly attracted by magnetic fields
- Permanently magnetized
- Example: Fe, Co, Ni etc.
Anti-Ferromagnetisms:
- Alignment of magnetic domains(group of metal ions) in the opposite direction of magnetic field
- Strongly repelled by magnetic fields
- Example: MnO
Ferrimagnetisms:
- Weakly attracted by magnetic fields
- Alignment of magnetic domains (group of metal ions) in the parallel and anti-parallel direction with direction of magnetic field and unequal in number.
- Loss magnetic moment on heating.
- Example: Fe3O4 (magnetite)