
Solids can be classified into three types on the basis of their conductivities.
Conductors:
The solids with conductivities ranging between 104 to 107 ohm–1m–1 are called conductors. Metals have conductivities in the order of 107 ohm–1m–1 is good conductors.
Insulators:
These are the solids with very low conductivities ranging between 10–20 to 10–10 ohm–1m–1.
Semiconductors:
These are the solids with conductivities in the intermediate range from 10–6 to 104 ohm–1m–1.
Conduction of Electricity
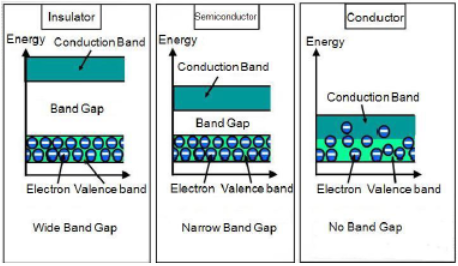
As we know that free electrons are responsible for the conduction of electricity in metals. Electrons are occupied in atomic orbital. The atomic orbital forms molecular orbital in metallic crystal. Molecular orbital are very closer in energy and known as bands. To be a conductor there must be some electrons in conduction band. But electrons are mainly occupied in valance band. On the basis of conductivity solid are classified in 3 types: Conductor, Semi-Conductor, and Insulator.
- In case of conductor there is an overlapping of valance band and conduction band.
- In semi-conductor there is small gap of energy between valance band and conduction band. Some of the electron may jump and show some activity. The metal showing this type of activity is known as intrinsic semi-conductors. For example: Silicon and Germanium
- In case of insulator there is a large energy difference between valance band and conduction band.
Classification of Semi-conductors:
The electrical conductivity of semi-conductors can be increased by adding some electron rich impurity or electron deficient impurity. This is called doping.
n-type semiconductor:
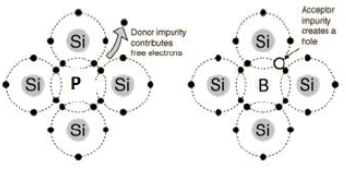
When the conductivity of semi-conductor is increased by adding electron rich impurity. It generates n-type semi-conductor.
For example: when silicon is doped with phosphorus, phosphorus also occupies some lattice site. The covalency of Si is 4 but that of P is 5. So, one electron per atom left unused and it delocalize from its location. This delocalizes electron help in increasing conductivity of semi-conductor.
p-type semiconductor:
When the conductivity of semi-conductor is increased by adding electron deficient impurity. It generates n-type semi-conductor.
For example: when silicon is doped with boron, boron also occupies some lattice site. The covalency of Si is 4 but that of B is 3. So, one of the positions of electron is left unused. This is called electron hole. The electron from neighboring atom moves to fill this hole but creates a new hole. This looks like movement of electron hole throughout the system. Electrons moves through theses hole under the influence of electric fields. In this way electrical conductivity increases.
Application of n-type and p-type semiconductors:
By the combination of n-type and p-type semiconductors many electronic devices are prepared.
- Diode (is a device that allows the current in one direction only) is used as amplifier.
- pnp and npn sandwiching is done in making transistors.
- As photo diode in solar cells.
- In lasers

