Nomenclature of Carbon Compounds
Nomenclature is the system of assignment of names given to organic compounds.
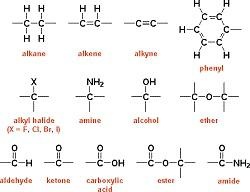
Heteroatom and Functional Group
- In hydrocarbon chain, one or more hydrogen atoms can be replaced by other atoms in accordance with their valencies. The element that replaces hydrogen is called a heteroatom.
- These heteroatoms and the group containing them impart chemical properties to the compound and hence are called functional groups.
Homologous Series
- It is a series of compounds in which the same functional group substitutes for hydrogen in a carbon chain. For instance, the Alcohols: CH2OH, C2H5OH,C3H7OH
- C4H9OH The successive member differs from one -CH2 unit and 14 units of mass.
- The chemical properties are imparted by the functional group thus all members have similar chemical properties. But the members have different physical properties. The physical properties vary among the members of homologous series due to a difference in their molecular mass.
- Melting point and boiling point increases with increasing molecular mass.