Paramagnetic substances are those which develop feeble magnetization in the direction of the magnetizing field. Such substances are feebly attracted by magnets and tend to move from weaker to stronger parts of a magnetic field.
Properties of paramagnetic substances:
Paramagnetic substances are those in which each atom or molecule has a net non-zero magnetic moment of its own:
- Susceptibility has a low positive value. (For example : χm for aluminum is +0.00002).
- Susceptibility is inversely proportional to absolute temperature (i.e.) χm α 1/T. As the temperature increases susceptibility decreases.
- The relative permeability is greater than one.
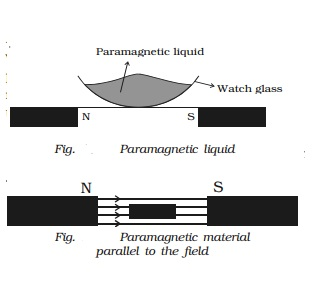
- When placed in a non uniform magnetic field, they have a tendency to move from weaker part to the stronger part of the field. They get magnetized in the direction of the field as shown in Fig.
- When suspended freely in a uniform magnetic field, they set themselves parallel to the direction of magnetic field (Fig.) Examples: Al, Pt, Cr, O2, Mn, CuSO4 etc.

