
Polyhalogen Compounds
Carbon compounds containing more than one halogen atom are known as polyhalogen compounds. For example: CFCl3, CH2Cl2 etc.
Some important polyhalogen compounds are described below:
Dichloromethane, CH2Cl2
Preparation of Dichloromethane:
Dichloromethane is prepared by the direct chlorination of CH4.
![]()
Properties of Dichloromethane:
- It is a colourless, sweet-smelling, volatile liquid.
- It has a low boiling point and low inflammability.
- It is insoluble in water
Uses of Dichloromethane:
- It is widely used as a paint remover.
- It is used as a solvent and cleaning agent in chemical manufacture, textiles, electronics, metals and plastic industries.
- It is also used as a process solvent in the manufacture of drugs.
Trichloromethane, CHCl3 (Chloroform)
Preparation of Trichloromethane:
CHCl3 is manufactured by chlorination of methane followed by separation by fractional distillation.
![]()
Properties of Trichloromethane:
- Chloroform is a sweet – smelling liquid.
- Chloroform is slowly oxidised by air in the presence of light to form poisonous phosgene gas

To avoid this oxidation chlorofrom is always stored in dark coloured bottles filled to the brim to exclude any air. Further bottles are also filled with small amount of ethyl alcohol to destroy traces of phosgene if formed, to harmless diethyl carbonate.
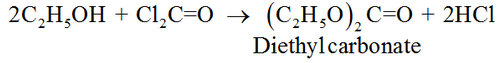
Uses of Trichloromethane:
- Chloroform is employed as a solvent for fats, alkaloids, iodine and other substances.
- It mainly used in the production of the freon refrigerant R-22.
Triiodomethane, CHI3 (Iodoform)
Preparation: Any compound containing CH3CO− or CH3CH(OH)− group, when heated with iodine and aquous NaOH gives yellow precipitate of iodoform.The reaction is known as iodoforin reaction.

Iodoform can also be prepared conveniently by using bleaching powder, CaOCl2.
![]()
Iodoform was used earlier as an antiseptic due to the liberation of free iodine but due to its objectionable smell, it has been replaced by other formulations containing iodine.
Tetrachloromethane, CCl4 (Carbon tetrachloride)
Preparation of Tetrachloromethane:
Cl4 is prepared by reacting carbon disulphide (CS2) with Cl2 in the presence of AlCl3.

Properties of Tetrachloromethane:
- Iodoform is a colourless, non-inflammable, poisonous liquid.
- When heated with steam at 773 K, it undergoes oxidation to form phosgene, COCl2 gas.
![]()
Uses of Tetrachloromethane:
- Carbon tetrachloride is used as a solvent for oils, fats, resins.
- It is used as a refrigerant and a dry-cleaning agent.
- It is used as a fire extinguisher under the name pyrene.
Freons
Preparation of Freons:
Dichloro difluoro methane or Freon 12 (CCl2F2) is one of the most common freons in and is manufactured from tetrachloromethane by Swarts reaction.
Properties of Freons:
- Freons are extremely stable, unreactive, non-toxic, non-corrosive and easily liquefiable gases.
- They lead to the depletion of ozone layer surrounding our planet.
Uses of Freons:
- Freons are used in aerosol propellants, refrigeration and air conditioning purposes.
Dichloro diphenyl trichloroethane (DDT)
Preparation of Dichloro diphenyl trichloroethane:
It is manufactured by the condensation of chloral with chlorobenzene in the presence of H2SO4.
Properties of Dichloro diphenyl trichloroethane:
- DDT is a white powder insoluble in water.
- It is a highly stable fat-soluble compound.
Uses of Dichloro diphenyl trichloroethane:
- It is mainly used as an insecticide.

