
Potassium Permanganate, KMnO4
Preparation of Potassium Permanganate:
It is prepared by fusion of MnO4 with alkali metal hydroxide (KOH) in presence of O2 or oxidising agent like KNO3. It produces dark green colored compound, K2MnO4 which undergoes oxidation as well as reduction in neutral or acidic solution to give permanganate.
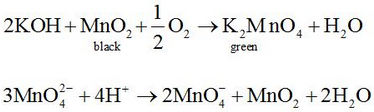
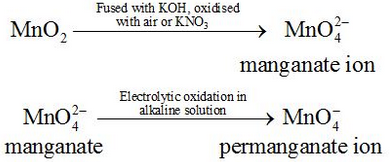
Commercially it is prepared by the alkaline oxidative fusion of MnO2 followed by the electrolytic oxidation of manganate (Vl).
Uses of Potassium Permanganate:
Potassium permanganate acts as a strong oxidizing agent in acidic, neutral or faintly basic medium.


