Trends in the M2+/M Standard Electrode Potentials
- There is no regular trend in the E0 (Mn2+/M) values. This is because their ionization enthalpies (I1 + IE2) and sublimation enthalpies do not show any regular trend.
- The general trend towards less negative E0 values along the series is due to the general increase in the sum of first and second ionization enthalpies.
- Copper shows a unique behaviour in the series as it is the only metal having positive value for E0. This explains why is does not liberate H2 gas from acids. It reacts only with the oxidizing acids (HNO3 and H2SO4) which are reduced. The reason for positive E0 value for copper is that the sum of enthalpies of sublimation and ionization is not balanced by hydration enthalpy.
- The values of E0 for Mn, Ni and Zn are more negative than expected from the general trend. This is due to greater stability of half-filled d-subshell (d5) in Mn2+, and completely filled d-subshell (d10) in Zn2+. The exceptional behaviour of Ni towards E0 value from the regular trend is due to its high negative enthalpy of hydration.
Trends in the M3+/M2+ Standard Electrode Potentials
- A very low value for E0 (Sc3+/Sc2+) reflects the stability of Sc3+ ion which has a noble gas configuration.
- The highest value for Zn is on account of very high stability of Zn2+ ion with d10 configuration. It is difficult to remove an electron from it to change it into +3 states.
- The comparatively high value of E0 (Mn3+/Mn2+) shows that Mn2+ is very stable which is on account of stable ds configuration of Mn2+.
- The comparatively low value of E0 (Fe3+/Fe2+) is on account of extra stability of Fe3+ (ds), i.e., low third ionization enthalpy of Fe.
- The comparatively low value for V is on account of the stability of V2+ ion due to its half-filled t32g configuration.
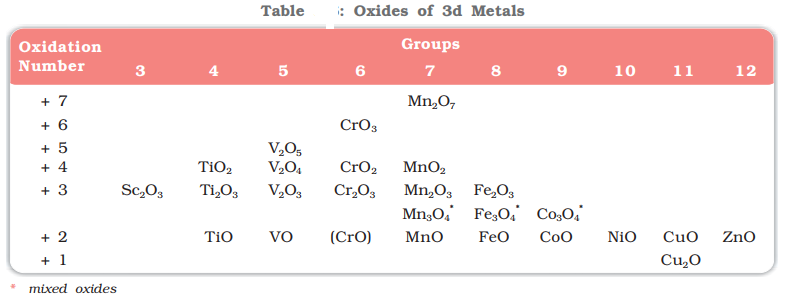
Trends in Stability of Higher Oxidation States