
Stratosphere:
Above the troposphere, between 10 and 50 km above sea level lies stratosphere. Ozone layer is one of the important constituents of stratosphere. The presence of ozone in the stratosphere prevents about 99.5 per cent of the sun’s harmful ultraviolet (UV) radiations from reaching the earth’s surface and thereby protecting humans and other animals from its effect.
Ozone Hole:
Depletion of ozone layer is known as ozone hole.
Formation of Ozone:
Ozone in the stratosphere is produced by UV radiations. When UV – radiations act on dioxygen (02) molecules, Ozone is produced.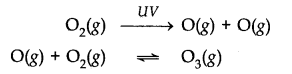
Ozone is thermodynamically unstable and decomposes to molecular oxygen. Thus, there exists an equilibrium between production and decomposition of Ozone molecules.
Depletion of Ozone layer:
Ozone blanket in the upper atmosphere prevent the harmful UV radiations from reaching earth.
But in recent years, there have been reports of depletion of this layer due to presence of certain chemicals in the stratosphere. Chlorofluorocarbons (CFCs), nitrogen oxides, chloride, CCl4 etc. are the chemicals responsible for depletion.
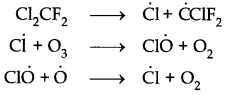
Chlorofluorocarbons dissociate in the presence of light gives chlorine free radicals which catalyze the conversion of ozone into oxygen.
Effects of the depletion of ozone layer:
With the depletion of ozone layer, more UV radiation filters into troposphere. UV radiations lead to ageing of skin, cataract, sunburn, skin cancer, killing of many phytoplanktons, damage to fish productivity etc.

