
Types of Polymerisation Reactions:
There are two broad types of polymerisation reactions, i.e., the addition or chain growth polymerisation and condensation or step growth polymerisation.
Addition Polymerisation or Chain Growth Polymerisation:
This mode of polymerisation leading to an increase in chain length or chain growth can take place through the formation of either free radicals or ionic species. However, the free radical governed addition or chain growth polymerisation is the most common mode.
Free radical mechanism:
A variety of alkenes or dienes and their derivatives are polymerized in the presence of a free radical generating initiator (catalyst) like benzoyl peroxide, acetyl peroxide, tert-butyl peroxide, etc.
- Chain initiating step: The process starts with the addition of phenyl free radical formed by the peroxide to the ethene double bond thus generating a new and larger free radical. This step is called chain initiating step.
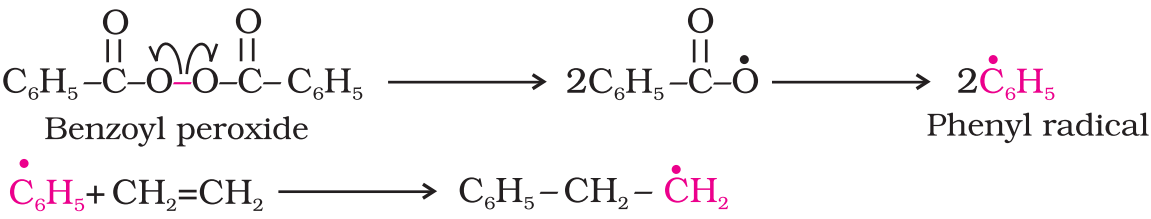
- Chain propagating step: The repetition of this sequence with new and bigger radicals carries the reaction forward and the step is termed as chain propagating step.

- Chain terminating step: Ultimately, at some stage the product radical thus formed reacts with another radical to form the polymerized product. This step is called the chain terminating step.
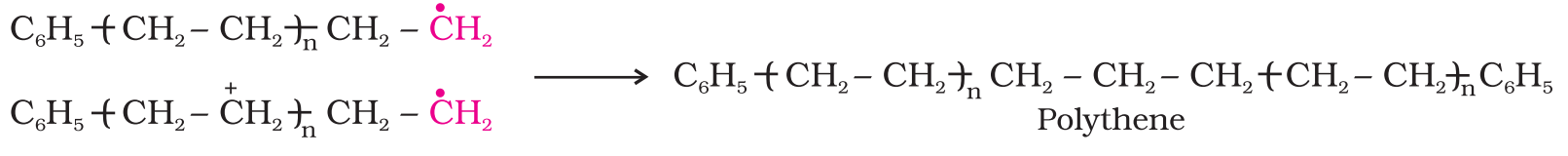
Preparation of some important addition polymers
Polythene:
There are two types of polythene as given below:
- Low-density polythene: It is manufactured by heating ethylene to 350-570k under pressure of 1000-2000 atmosphere and in presence of a trace of oxygen and peroxide
Properties: Low-density polyethene is a transparent polymer of moderate tensile strength and high toughness. It is a poor conductor of electricity.
Uses: It is used as packaging material and in the production of wires and cables, squeeze bottles. - High-density polyethene: In the process of preparation ethane is heated to 333-343k under a pressure of 6-7 atmospheres in presence of a catalyst consisting of triethyl aluminium and titanium trichloride and tetrachloride.
Polyterafluoroethane:
It is manufactured by heat tetrafluoroethene in presence of peroxide or ammonium persulphate catalyst at high pressures.

Uses: It is Flexible and inert to solvents and to boiling acids. Because of its great chemivalintertness and high thermal stability, it is used for making non-stick utensils.
Polyacrylonitrile:
Starting material acrylonitrile (CH2=CH-CN)
Reaction: Addition polymerisation of acrylonitrile in presence of peroxide catalyst gives polyacrylonitrile.
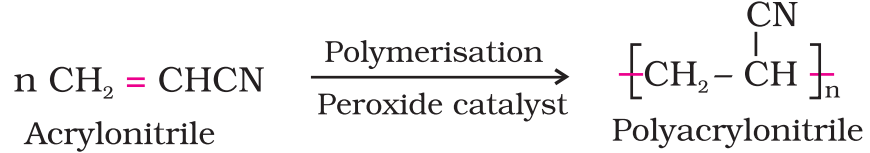
It is a hard horny and melting material.
Uses:
- It is used as a substitute for wool in the manufacture of Orion and Acrilan fibres.
- It is also used in the preparation of other polymers.

