
Group (1 & 2) belong to the s-block of the Periodic Table.
- Group 1 consists of: lithium, sodium, potassium, rubidium, caesium and francium and collectively known as the alkali metals.
- Group 2 include: beryllium, magnesium, calcium, strontium, barium and radium.
Except Beryllium they are known as alkaline.
Group 1 Elements: Alkali Metals
Lithium, Sodium, Potassium, Rubidium, Caesium and Francium
Physical Properties Alkali Metals:
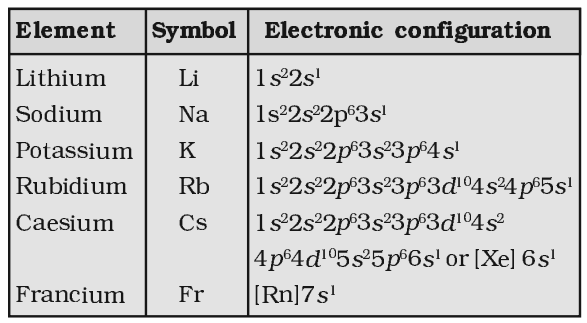
Electronic Configuration:
[noble gas] ns1, where n represents the valence shell.
Atomic and Ionic Radii
- Atomic and ionic radii of alkali metals increase on moving down the group i.e., they increase in size going from Li to Cs.
- Alkali metals form monovalent cations by losing on valence electron. Thus, cationic radius is less as compared to the parent atom.
Ionization Enthalpy
- The ionization enthalpies of the alkali metals are generally low and decrease down the group from Li to Cs.
- This is because the effect of increasing size outweighs the increasing nuclear charge, and the outermost electron is very well screened from the nuclear charge.
Hydration Enthalpy
- The hydration enthalpies of alkali metal ions decrease with increase in ionic sizes.
![]()
- Li+ has maximum degree of hydration and for this reason lithium salts are mostly hydrated
Metallic Radius:
The metallic character increases down the group.
Melting point and boiling point:
The m.p. and b.p. of alkali metals are very low and decrease with increase in atomic number.
Nature of bonds formed:
These metals form ionic bonds. The ionic character increases as we down the group.
Flame colouration:
All the alkali metals impart a characteristic colour to the flame.
Photoelectric effect:
Alkali metals (except Li) exhibits photoelectric effect.
Oxidation state:
The alkali metals exhibit oxidation state of +1 in their compounds and are strongly electropositive in character. The electropositive character increases from Li to Cs.

Chemical Properties of Alkali
The alkali metals are highly reactive due to their large size and low ionization enthalpy. The reactivity of these metals increases down the group.
Reactivity towards air:
On exposure to moist air, their surface get tarnished due to the formation of their oxides, hydroxides and carbonates.
![]()
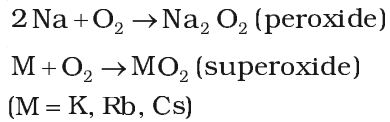
Hence, they are kept under inert liquid like kerosene oil but lithium is kept wrapped in paraffin wax because it floats on the surface of kerosene oil due to its low density.
Reactivity towards water:
Alkali metals react with water and other compounds containing acidic hydrogen atoms such as hydrogen halides, acetylene etc. to liberate hydrogen gas.
![]()
Reactivity towards dihydrogen:
Alkali metals react with dry hydrogen at about 673 K to form crystalline hydrides which are ionic in nature and have high melting points.
![]()
Reactivity towards halogens:
The members of the family combine with halogen to form corresponding halides which are ionic crystalline solids.
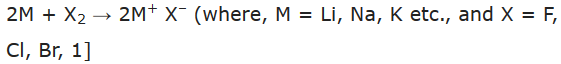
Reactivity of alkali metals with particular halogen increases from Li to Cs.
Reducing nature:
The alkali metals are strong reducing agents. In aqueous solution it has been observed that the reducing character of alkali metals follows the sequence Na < K < Rb < Cs < Li, Li is the strongest while sodium is least powerful reducing agent. This can be explained in terms of electrode potentials (Eo). Since the electrode potential of Li is the lowest.

Solutions in liquid ammonia:
All alkali metals dissolve in liquid ammonia giving deep blue solutions which are conducting in nature.
![]()
When light falls on the ammoniated electrons, they absorb energy corresponding to red colour and the light which emit from it has blue colour.
In concentrated solution colour changes from blue to bronze. The blue solutions are paramagnetic while the concentrated solutions are diamagnetic.
Uses of Alkali Metals
- Lithium is used as deoxidizer in the purification of copper and nickel.
- Lithium is used to make both primary and secondary batteries.
- Lithium hydride is used as source of hydrogen for meteorological purposes.
- Used as sodium amalgum in laboratory (synthesis of organic compounds).
- Sodium is used in sodium vapour lamp.
- Salts of potassium are used in fertilizers, reducing agent.
- In Cesium, rocket propellent and photographic cells.
Anomalous Properties of Lithium
- Exceptionally small size of its atom and ion.
- High polarizing power (i.e., charge/radius ratio).
- As a result, there is increased covalent character of lithium compounds which is responsible for their solubility in organic solvents.
Diagonal relationship between Li and Al
Li resembles Mg mainly due to similarity in sizes of their atoms and ions.
The main points of similarity are:
- Both are quite hard.
- Both LiOH and Mg(OH)2 are weak bases.
- Carbonates of both on heating decompose to produce oxides and carbondioxide.
- Both react with nitrogen to give ionic nitrides.
- Nitrates of both decompose on heating to give oxides.
- Both Li and Mg do not form solid bicarbonates.
- Because of covalent character LiCl and MgCl2 are soluble in enthanol.
- The hydroxides, bicarbonates and fluorides of both Li and Mg are sparingly soluble in water.
Biological importance of Na and K
- Sodium ions participate in the transmission of nerve signals.
- Sodium ions also regulate flow of water across the cell membranes and in transport of sugars and amino acids into the cells.
- Potassium ions are the most abundant cations within cell fluids, where they activate many enzymes, participate in oxidation of glucose to produce ATP.
- Potassium ions in combination with sodium ions are responsible for transmission of nerve signals.
- The functional features of nerve cells depend upon the sodium potassium ion gradient that is established in the cell.

