
Alkaline Earth Metals:
They were named alkaline earth metals since they were alkaline in nature like alkali metals oxides and they were found in the earth’s crust.
Example: Be (Beryllium), Ca, Mg, Sr, etc.
Physical Properties of alkaline earth metals
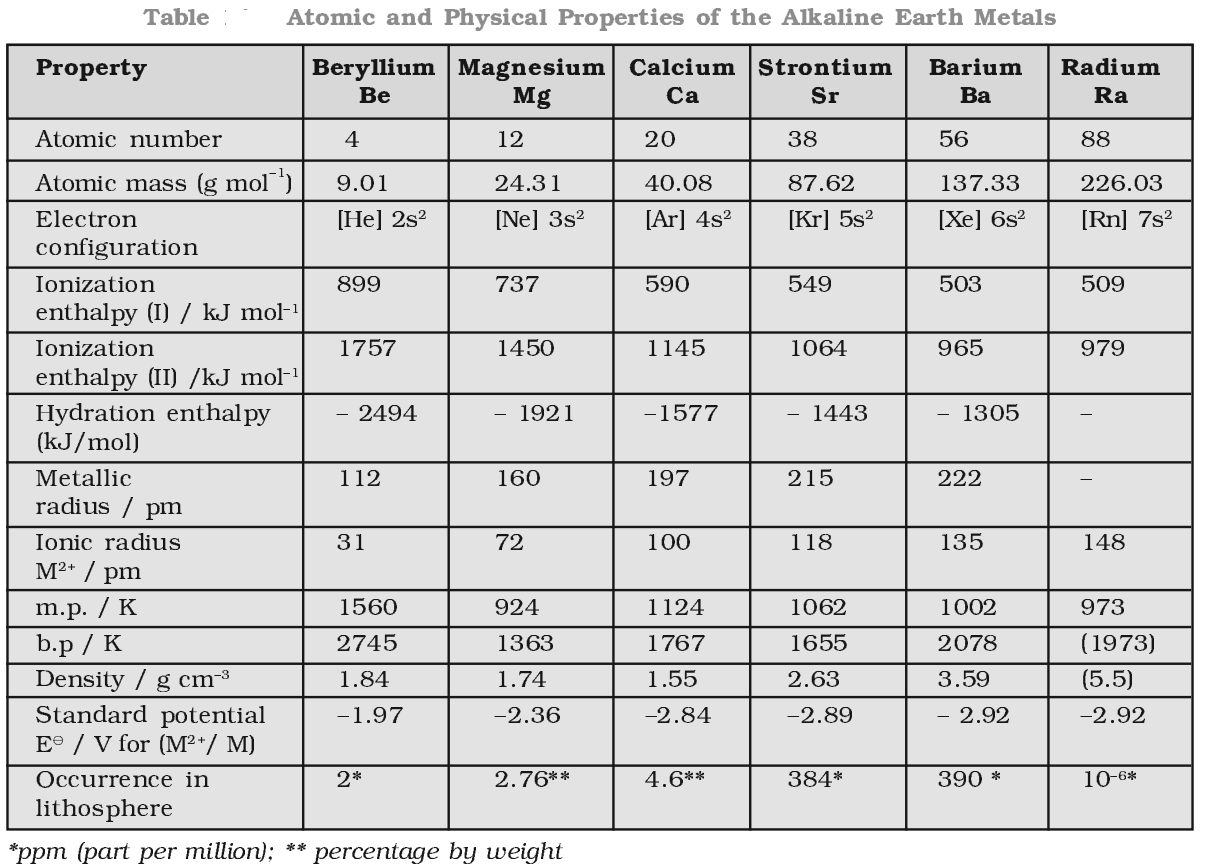
Electronic configuration of alkaline earth metals
[noble gas] ns2
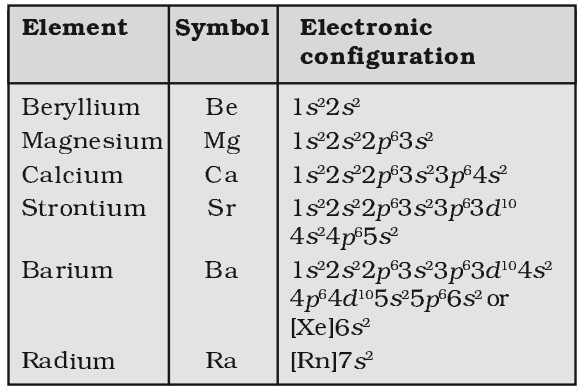
Atomic and Ionic Radii of alkaline earth metals:
The atomic radii and ionic radii of these elements are quite large but smaller than those of the corresponding alkali metals. Due to increased nuclear charge of these elements. The atomic as well as ionic radii go on increasing down the group due to the gradual addition of extra energy level.
Ionization enthalpy of alkaline earth metals:
The alkaline earth metals have fairly low ionization enthalpies though greater than those of the corresponding elements of group 1 and this value decreases down the group.
Oxidation State of alkaline earth metals:
All the members of the family exhibit +2 oxidation state in their compounded and the form divalent cations (M2+)
Metallic Character of alkaline earth metals:
Alkaline earth metals have stronger metallic bonds as compared to the alkali metals present in the same period.
Electronegativity of alkaline earth metals:
The electro negativity values of alkaline earth metals are quite close to those of alkali metals, though slightly more.
Melting and boiling point of alkaline earth metals:
The melting point and boiling points of these metals are higher than those of alkali metals present in the same period.
Density of alkaline earth metals:
These are much denser than alkali metal is because of their smaller size and greater nuclear charge and mass. The density however, first decreases from Be to Ca and then steadily increase from Ca to Ra due to difference in type of crystal structure.
Reducing character of alkaline earth metals:
Alkaline earth metals are weak reducing agent than the corresponding alkali metals which have lower ionization enthalpies and comparatively bigger atomic sizes.
Chemical Properties of alkaline earth metals:
The alkaline earth metals are less reactive than the alkali metals. The reactivity of these elements increases on going down the group.
Reactivity towards water:
Since these metals are less electropositive than alkali metals, they are less reactive towards water.
![]()
Magnesium reacts with boiling water or steam. Rest of the members reacts even with cold water.
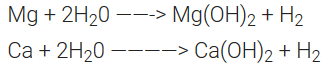
Reaction towards oxygen:
The affinity towards oxygen increases down the group. Thus, Be, Mg and Ca when heated with O2 form monoxides while Sr, Ba and Ra form peroxides.


Reactivity towards the halogens:
They combine with the halogens at appropriate temperature to form corresponding halides MX2.
M + X2 MX2 (X = F, Cl, Br, I)
Thermal decomposition of (NH4)2 BeF4 is used for the preparation of BeF2.
Reactivity towards hydrogen:
These metals except Be combine with hydrogen directly upon heating to form metal hydrides.
![]()
![]()
Reactivity towards acids:
Alkaline earth metals except Be, displace H2 from acids.
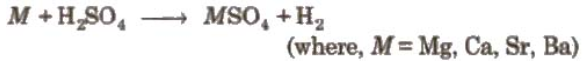
Reactivity increases down the group from Mg to Ba. Only Mg displaces H2 from a very dilute HNO3.
Solutions in liquid ammonia:
Like Alkali Metals, these metals also dissolve in liquid ammonia by giving coloured solutions.
![]()
The tendency to form ammoniates decreases with increase in size of the metal atom (i.e., on moving down the group).
Uses of Some important compounds
Caustic soda:
- It is used in Soap, paper, textile, petroleum industry.
Sodium carbonate
- It is used in glass and soap industry.
- In metal refining.
- In production of sodium compounds such as borax, caustic soda, sodium phosphate etc.
- In paper making and textile manufacturing.
Quick lime
- It is used in the preparation of cement, glass and calcium carbide.
- In the purification of sugar.
- In softening of hard water.
- As a flux in the extraction of mental.
Lime stone
- It is used as building material.
- In the manufacture of quick lime.
- In Solvay process to prepare Na2CO3 as it is a source of CO2.
- In toothpaste and certain cosmetics.
Cement:
- It is an important building material. It is used in concrete and reinforced concrete, in plastering and in the construction of bridges, dams and buildings.
Plaster of paris
- It is used in making for pottery and ceramics etc.
- In surgical bandages for setting broken bones of the body.
- For making statues, models, decorative materials and black board chalk.
General Characteristics of Compounds of Alkaline Earth Metals
Oxides and Hydroxides
- The alkaline earth metals bum in oxygen to form MO (mmonoxide).
- These oxides are very stable to heat.
- Hydroxides of alkaline earth metals are less stable and less basic than alkali metal hydroxides.
- Beryllium hydroxide is amphoteric in nature.
Halides
- The alkaline earth metals combine directly with halogens at appropriate temperatures forming halides, MX2. They can also be prepared by the action of halogen acids (HX) on metals, metal oxides, metal hydroxides.

- Except beryllium halides, all other halides of alkaline earth metals are ionic in nature.
- Except BeCl2 and MgCl2 other chloride of alkaline earth metals impart characteristic colours to flame.
![]()
- The tendency to form halide hydrates decreases down the group.
![]()
Salts of Oxoacids:
The alkaline earth metals also form salts of oxoacids. Some of these are:
Carbonates:
- Carbonates of alkaline earth metals are thermally unstable and decompose on heating.
- The thermal stability increases from BeCO3 to BaCO3.
- BeCO3 is unstable and kept only in the atmosphere of CO2.
- Solubility in water: BeCO3 is highly soluble in water whereas BaCO3 is almost insoluble.
Nitrates:
- Nitrates of alkaline earth metals are prepared by treating the corresponding metal carbonates with dilute HNO3.
For example:

(M = Be, Mg, Ca, Sr, Ba)
- All these metal nitrates decompose on heating to give the oxide.
For example:
![]()
(M = Be, Mg, Ca, Sr, Ba)
Sulphates:
- The sulphates of alkaline earth metals are white solids and quite stable to heat.
- BeSO4 and MgSO4 are readily soluble in water. Solubility decreases from BeSO4 to BaSO4.
Reason: Due to greater hydration enthalpies of Be2+ ions and Mg2+ ions they overcome the lattice enthalpy factor. Their sulphates are soluble in water.
Anomalous Behavior of Beryllium
Beryllium, differs from the rest of the members of its group due to the following reasons:
- Beryllium has a small atomic and ionic size.
- It has no vacant d-orbitals.
- It has a high charge density
Diagonal Relationship Between Be and Al
The main identical physical and chemical properties of Be with aluminum are given below
- Action of air: Both the metals are stable in air.
- Action with water: Be and Al do not decompose water even at 373K. It is due to their less electropositive character.
- Electropositive character: Beryllium like aluminum is less electropositive due to their small ionic radii.
- Complex formation: Beryllium and aluminum form a number of complexes. Both form fluoro complex anions like BeF2-4 and AlF3-6 in solution.
- Reaction with alkali: Beryllium and aluminum react with sodium hydroxide liberating hydrogen.
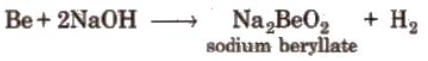
- Amphoteric character of oxides: Oxides of both Be and Al are amphoteric in nature. So, they dissolve both in acids as well as in alkalies.
Biological importance of Ca and Mg
- Magnesium ions are concentrated in animal cells and Calcium ions are concentrated in body fluids, outside the cell.
- All enzymes that utilize ATP in phosphate transfer require magnesium ion as cofactor.
- In green plants magnesium is present in chlorophyll.
- Calcium and magnesium ions are also essential for the transmission of impulses along nerve fibers.
- Calcium ions are important in blood clotting and are required to trigger the contraction of muscles.
- Calcium ions also regulate the beating of the heart.

