
It is the study of the flow of energy. It encompasses the study of the flow of heat as well as mass.
Thermodynamics terminology
System: The part of the universe observation is called system.
Surrounding: The part of the Universe not under observation is called Surrounding.

System + Surrounding = Universe
Types of System
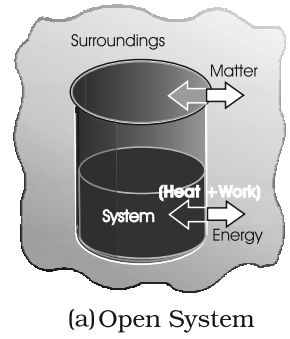
- Open System: A system in which both flow of mass and heat is possible.
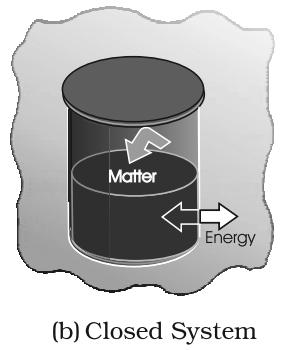
- Closed System: A system in which flow of heat is possible but flow of mass is not possible.
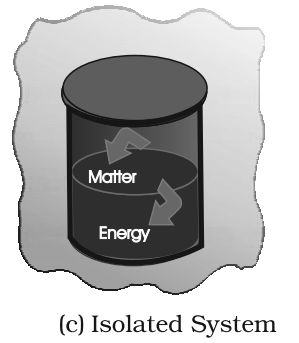
- Isolated System: A system in which neither heat nor mass can flow in or out.
The State of the System
State of System: When microscopic properties have definite value, the conditions of existence of the system is known as the state of the system.
State function (State variables): When values of a system is independent of path followed and depend only on initial and final state, it is known as state function, e.g., ∆U, ∆H, ∆G etc.
Path function: These depend upon the path followed, e.g., work, heat, etc.

